The medical device industry faces continuous pressure for faster imaging, smaller instruments, greater reliability, and stricter regulatory compliance. To the OEMs producing next-generation endoscopy systems, robotic surgical instruments or a new generation ultrasound platform, the cable assembly embedded in the device is no longer a passive component. It is a performance, patient safety and commercial success determinant.
Hotten Electronic Wire focuses on OEM micro coax cable assemblies to be used with manufacturers of medical devices. Our production facility covers 10,000 square meters and has 40 high-technology manufacturing units and a yearly production of more than 144 million meters, we are not just a supplier, but a strategic continuation of the engineering and supply chain sections of our customers.
The OEM Challenge: Why Standard Cables Are Not Enough
There is a paradox facing medical device OEMs. The miniaturization requirement is driving the cable diameters to the lowest levels but clinical expectations of the resolution of an image and diagnostic accuracy are on the increase. Ready-made cable assemblies, particularly those designed for consumer electronics or a general-purpose industrial device, cannot meet these requirements.
The signal integrity of high frequencies, very high flexibility, sterilization, and biocompatibility have to coexist in a diameter that is usually within fractions of a millimeter. In addition, these requirements differ between applications. A disposable bronchoscope does not face the same lifecycle requirements as a reusable surgical arthroscope. Echocardiography probe used transesophageally needs dissimilar mechanical capacities as compared to a handheld ultrasound probe.
That is why OEMs are more willing to find custom-engineered solutions instead of catalog products. At Hotten, every OEM engagement begins not by a price quote, but by a technical discovery.
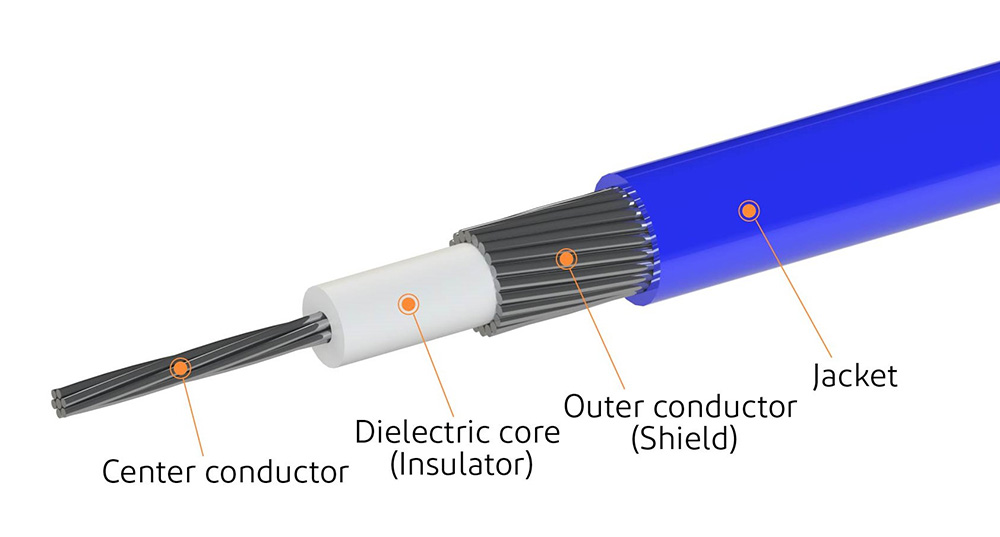
Our OEM Approach: Engineering as the Starting Point
When the manufacturer of a medical device wants to work with Hotten to assemble a micro coax cable, we do not ask for the part number. We ask:
What are the signal frequency and bandwidth requirements?
Mechanical constraints: bend radius, flex life, insertion force?
Which sterilization methods will the device undergo?
What production volume is expected, prototype, pilot, full-scale start-up?
What are the regulatory requirements- FDA 510(k), MDR or PMA?
These are the questions that lead to any subsequent decision made: stranding of conductors, dielectric material, shielding layout, the jacket polymer, connector choice, and the termination requirements.
Case Example: Miniaturizing a Surgical Camera Cable
One of the more recent partnerships that we have entered into with a major OEM of robotic-assisted surgery platforms exemplifies our OEM philosophy. The client needed a very thin coax assembly with the ability to transfer uncompressed 4K video over a 5mm instrument shaft- and still be autoclave compatible.
Coax assemblies of standard 50AWG were too thick. Available 52AWG products in the market did not have the mechanical stability of 1,000 or more sterilization cycles. Connectors off-the-shelf introduced impedance mismatches that reduced the image quality.
The design of a custom solution was by the R&D team of Hotten, which develops more than 300 new cable designs in a single year:
A conductor made of silver-plated copper, 54AWG and minimizing signal loss while reducing diameter.
Micro-porous PTFE dielectric with 50 Ω impedance in a 0.28mm profile.
100% spiral-wrapped foil with ultra-fine braid dual-layer shielding.
A proprietary polyether ether ketone (PEEK) jacket which has been tested at 1,200 cycles of the autoclave without cracking or discoloring.
Strain relief molded to fit perfectly round the sterile interface of the OEM.
The result? A cable assembly allowing the OEM to cut instrument shaft diameter by 18 percent and increase signal-to-noise ratio by 3.2dB, providing a measurable competitive advantage and delivered in 14 weeks after concept approval.

Regulatory Compliance: Built-In, Not Bolted-On
In the case of medical device OEMs, component sourcing is a risk to regulation. When a cable assembly fails a biocompatibility test, or fails in sterilization validation testing, the whole device submission may be delayed - months and millions of dollars.
Hotten solves this by using design-for-compliance engineering. We do not view regulatory requirements as an end verification point but we incorporate them in the process design and material selection.
Our medical cable assemblies are OEM and are designed with:
Material selection (cytotoxicity, sensitization, irritation) in accordance with ISO 10993.
Autoclave, EtO, gamma, compatibility with electron-beam sterilization.
Prop 65, REACH, and RoHS control of the restricted substances.
Complete material traceability used in harnesses, starting with resin lot and ending with finished harness.
Documentation of design verification to OEM design verification Design History File (DHF) documentation Design History File (DHF) documentation supporting OEM design verification.
To clients seeking FDA or CE marking, we supply technical dossiers, process validation Documentation and supplier statements which can be incorporated directly into the regulatory submissions of clients.
Scalability: From Engineering Samples to High-Volume Production
OEMs have to contend with transitioning between prototype and production as one of the most frequent challenges. A cable assembly carefully hand-terminated in an engineering laboratory might be impossible to scale to large-scale production may have unacceptable yield loss on transfer to large scale production.
The manufacturing engineering team at Hotten plays this role. We do not have generic assembly lines in our 40 production units, they are set up and equipped to do micro-coax-specific operations, and these include:
Laser stripping machines that do not nick the conductor or dielectric layers.
Ultra-miniature connector calibrated semi-automatic termination presses.
Vision-inspection stations to check concentricity, solder fillet quality and contact positioning.
Not only first article impedance profiling but impedance profiling of all production lots.
We can scale our engineering samples of 50 pieces all the way up to 50,000-unit production orders with this infrastructure and not requalify or performance drift.
Supply Chain Integration: JIT, Kanban, and Global Logistics
The schedules of medical devices production are highly variable. The enrollment of the clinical trials varies. Approaches to regulatory approvals are either proactive or reactive. The peaks of demand in consumable devices are caused by seasonal volumes of procedures.
Being an OEM partner, Hotten will synchronize our production plans and inventory needs with the demand indicators of our clients. We support:
Just-in-time (JIT) delivery based on OEM assembly lines.
Kanban replenishment systems of high usage components.
Consignment inventory projects at customer designated logistic hubs.
Shipping to North American, European and Southeast Asian contract manufacturers by drop-ship.
We have an annual production capacity of more than 144 million meters of cable that allows us to supply even when our clients grow in their product introduction to their market dominant position.
Confidentiality and IP Protection
The architecture of products and relationships with supply chains are the proprietary assets in the competitive medical devices landscape. Hotten signs extensive Non-Disclosure Agreement (NDAs) with all of its OEM partners and has strict access controls in our engineering and production area.
We do not take customer-specific designs and use them with other customers, or do we post recognizable OEM project information in our marketing texts without express permission. We are an established supplier of OEMs with a reputation of technical ability, but also discretion and integrity.
Conclusion: More Than a Vendor
The term OEM supplier implies a transactional relationship: specification made, components received, and money is paid. At Hotten we do not accept that restriction.
We consider ourselves as a part of the engineering, quality and manufacturing organizations of our customers. By partnering with Hotten, a medical device OEM can access dedicated skills in micro-coax domain, regulatory acumen, and affordable scale of production; all in a single relationship.
That integration is no longer optional in an industry where competitors are measured by miniaturization, which is the competitive advantage, and regulators by compliance, which is the competitive entrance to a market. It is a strategic necessity.
When designing a medical device that requires a high frequency signal integrity in a miniature-scale package, contact Hotten Electronic Wire. Together, we can create your advantage.
 Hot News
Hot News2025-12-17
2025-12-11
2025-12-05
2025-04-29