ဆေးဘက်ဆိုင်ရာပစ္စည်းများအတွက် မိုက်ခရိုကော့စ်ကြိုးအစီအစဉ်များသည် လျှပ်စစ်၊ ယန္တရားနှင့် စည်းမျဉ်းစည်းကမ်းဆိုင်ရာ လိုအပ်ချက်များကို အထူးတွင် တင်းကြပ်စွာ ဖော်ထုတ်ရမည်။
ဆေးဘက်ဆိုင်ရာပစ္စည်းများ ဖွံ့ဖြိုးရေးအတွင်း အရေးအကြီးဆုံး ရွေးချယ်မှုများထဲမှ တစ်ခုမှာ မိုက်ခရိုကော့စ်ကြိုးအစီအစဉ်များကို သင့်လျော်စွာ ရွေးချယ်ရေးဖြစ်သည်။ အင်တာကွန်နက် (interconnect) ဖြေရှင်းနည်းသည် ပုံရိပ်ဖမ်းယူမှုအရည်အသွေး၊ ပစ္စည်း၏ လှုပ်ရှားမှုလွယ်ကူမှုနှင့် လူနာအား အန္တရာယ်ကင်းမှုတို့ပေါ်တွင် တိုက်ရိုက်သက်ရောက်မှုရှိသည်။ မည်သည့်အမျိုးအစားဖြစ်စေ အော့လ်ထရိုင်စ် ပရိုဗီး ကেဘော့ ၊ အင်ဒိုစကော်ကျူအလိုင်း ၊ IVUS Catheter သို့မဟုတ် ခွဲစိတ် Scalpel Cable . ဆေးဘက်ဆိုင်ရာ ကိရိယာ အင်ဂျင်နီယာ သန်းချီရှိပြီး ခက်ခဲတဲ့ သတ်မှတ်ချက် လုပ်ငန်းစဉ်တစ်ခုလုံးမှာ လမ်းညွှန်ဖို့ Hotten မှာ ကျွန်မတို့နဲ့အတူ အလုပ်လုပ်ခဲ့ကြတယ်။ အောက်ပါအချက်များသည် နည်းပညာသတ်မှတ်ချက်များနှင့် စည်းမျဉ်းစည်းကမ်းများနှင့် ကိုက်ညီသော micro coaxial cable များအတွက် သတ်မှတ်ချက်များအတွက် လက်တွေ့ကျသော လမ်းညွှန်ချက်များဖြစ်သည်။
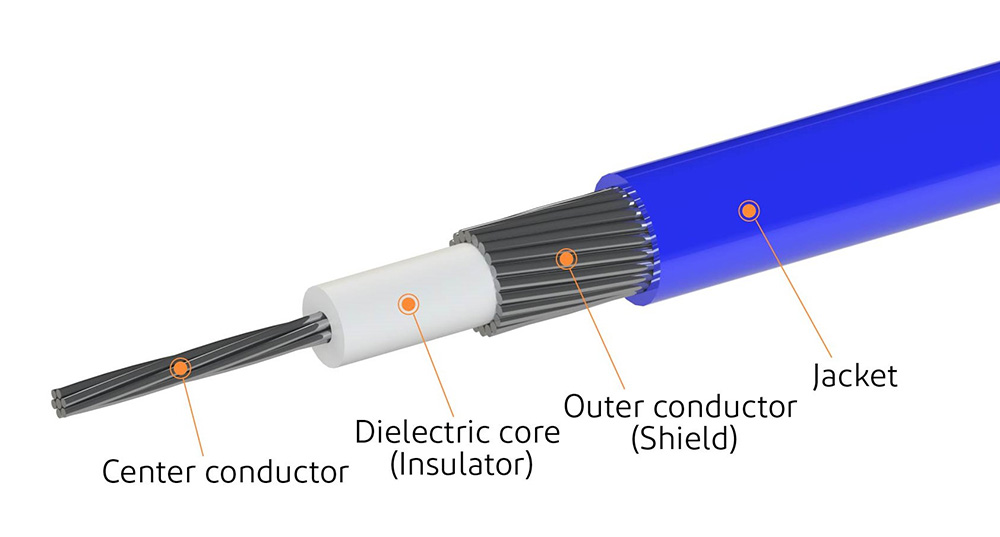
လျှပ်စစ်စွမ်းဆောင်ရည် လိုအပ်ချက်များကို သတ်မှတ်ခြင်း
အသေးစိတ်အချက်အလက်တွေကို သင်ရဲ့ အက်ပ်နဲ့ ဘာလုပ်ချင်လဲဆိုတာ သိခြင်းနဲ့ စပါတယ်။ အချက်ပြမှု တစ်သမတ်တည်းဖြစ်ခြင်းဟာ အမြန်ပုံထုတ်ခြင်းလိုမျိုးမှာ အရေးပါဆုံးပါ။ ICE ကေဘယ်လ်များ နှင့် Ultrasound Probe Cables များ . စပြီး ဆုံးဖြတ်လိုက်ပါ
အတားအဆီးလိုအပ်ချက်များ 50Ω သို့မဟုတ် 75Ω သရုပ်ဖော်မှု အင်ပီဒန်ဆန်မှုသည် ဆေးဘက်ဆိုင်ရာ ပုံထုတ်စနစ် အများစုတွင် သာမန်ဖြစ်သည်။ အညီအမျှ လုပ်ဆောင်မှုရှိစေရန် ခွင့်ပြုချက် (±3-5Ω သာမန်) ကို ဖော်ပြပါ။
ဘန်ဘဒ်အလျားနှင့် လျှော့ချမှု: ကြိမ်နှုန်းများ တိုးလာခြင်းသည် လမ်းညွှန်စက်၏ အကျယ်အဝန်းများ သို့မဟုတ် အထူးအပူထိန်းစက်များ တိုးလာရန် လိုအပ်သည်။ RF Ablation Cables များတွင် MHz များစွာဖြင့် အလုပ်လုပ်နေပါက လျှပ်စစ်လျှော့ချမှုသည် လုပ်ငန်းစဉ် အောင်မြင်မှုကို တိုက်ရိုက် သက်ရောက်မှုရှိသည်။
လျှပ်ကာပါဝါနှင့် အချက်ပြမှု မြင့်တက်လာချိန်များ- ဤပါရာမီတာနှစ်ခုသည် EEG လီဒ်ဝိုင်ယာများနှင့် သွားနှင့်ခြောက်ဆီးဆိုင်ရာ စိတ်ကူးယဉ်ခြင်းကြိုးများကဲ့သို့သော ကိရိယာများ၏ အချိန်မှန်ကန်မှုတိုင်းတာမှုများကို အကျိုးသက်ရောက်စေသည်။
ဤအသေးစိတ်အချက်အလက်များကို ကုန်စည်ဖောက်သည်နှင့် မျှဝေခြင်းဖြင့် ကိရိယာ၏ အဓိကဒီဇိုင်းသည် စမ်းသပ်မှု သို့မဟုတ် ကုသမှုလုပ်ဆောင်ချက်များကို အစပိုင်းတွင်ပဲ ထိရောက်စွာ အကောင်အထည်ဖော်နိုင်ရန် အာမခံပေးပါသည်။

ယန္တရားနှင့် အရွယ်အစားဆိုင်ရာ ကန့်သတ်ချက်များကို ဆုံးဖြတ်ပါ
ဆေးဘက်ဆိုင်ရာကိရိယာများသည် အထူးသဖြင့် အနည်းငယ်သာ ထိရောက်သော ကိရိယာများသည် အလွန်ကျဉ်းမောင်းသော နေရာများတွင် အလုပ်လုပ်ကြသည်။ သင့်၏ မိုက်ခရိုကော့စ်အစီအစဥ်၏ အရွယ်အစားသည် လူနေမှုနေရာတွင် လုပ်ဆောင်နိုင်မည့် အရာကို အများအားဖြင့် ဆုံးဖြတ်ပေးသည်။ အသေးစိတ်အချက်အလက်များကို သတ်မှတ်ရာတွင်-
စုစုပေါင်း အချင်းအရွယ်: အောက်စ်ကော့စ်ကြိုးများနှင့် IVUS ကက်သီတာများတွင် 0.1mm တစ်ခုစီသည် အရေးကြီးသည်။ 30-46 AWG အထိ အလွန်ပေါ့ပါးသော ကော့စ်ကြိုးများကို အသုံးပြု၍ 2-3mm အလုပ်လုပ်သော နေရာတွင် ပုံရေးပုံဖော်မှု အချိန်ပိုင်းများကို အများအားဖြင့် ပေးနိုင်ပါသည်။
အက်က်စ်ဖ်လက်စ် (Flex Life) လိုအပ်ချက်များ- နှလုံးခုန်နှင့် အဆစ်များ လှုပ်ရှားမှုတွင် အသုံးပြုရန် ပေါ့ပါးသော ကိရိယာများကို လိုအပ်ပါသည်။ မည်မျှသော အက်က်စ်ဖ်လက်စ် စက်ကွင်းများကို မျှော်လင့်ထားသည်ကို ဖော်ပြပါ။ အလွန်ကျဉ်းသော အက်က်စ်ဖ်လက်စ် အက်ဒီယပ်စ်များအတွက် အထူးသော ကြိုးများ၏ ဖွဲ့စည်းပုံများ လိုအပ်ပါသည်။
ဆွဲခြင်းအားနှင့် ဖိစိပ်မှုကို ခံနိုင်ရည်ရှိမှု - သွေးထွက်ခြင်းနှင့် အသက်ကာကွယ်ရေး လုပ်ထုံးလုပ်နည်းများတွင် အသုံးပြုသည့် ဆာဂီကယ် စကယ်ပယ် ကြိုးများနှင့် ရိုဘော့စ် ဝိုင်ယာ ဟာနီစ်များသည် အလွန်များပြားသော ဆွဲအားများကို ခံနိုင်ရည်ရှိရပါမည်။ ထို့ကြောင့် ကေဗလာ (Kevlar) သို့မဟုတ် စတီလ်သံမဏိ ပေါင်းစပ်မှုများကဲ့သို့သော အားကောင်းသည့် အစိတ်အပိုင်းများကို ထည့်သွင်းရန် အကောင်းဆုံးဖြစ်ပါသည်။
သင်၏ မော်ရ်ရှင်နှင့် ပတ်သက်သည့် လိုအပ်ချက်များကို ရှင်းလင်းစွာ ဖော်ပြပါ။ လိုအပ်ချက်များကို အလွန်အမင်း ဖော်ပြပါက စရိတ်များ ပိုများလာမည်ဖြစ်ပြီး၊ လိုအပ်ချက်များကို အလွန်နည်းပါးစွာ ဖော်ပြပါက သင်၏ စက်ကို အသုံးပျော့သွားမည်ဖြစ်ပါသည်။
ဇီဝသ совместимость (Biocompatibility) နှင့် သန့်စင်ခြင်း သဟဇាឤ (Sterilization Compatibility) ကို ဖော်ပြပါ
ဆေးဘက်ဆိုင်ရာ ကြိုးများအတွက် အမြင့်ဆုံး လိုအပ်ချက်ဖြစ်ပါသည်။ လူနာများနှင့် ထိတွေ့မှုရှိသည့် အတွက် အသုံးပြုရန် အထိအထိ စမ်းသပ်ပြီး ထပ်ခါထပ်ခါ သန့်စင်မှုကို ခံနိုင်ရည်ရှိသည့် ပစ္စည်းများကို အသုံးပြုရပါမည်။ သင်၏ အသေးစိတ်ဖော်ပြချက် စာရွက်တွင် ဖော်ပြပါ -
ISO 10993 စံနှုန်းများကို တိကျစွာ ဖော်ပြပါ - လူနာနှင့် ထိတွေ့မှုရှိသည့် ပစ္စည်းအားလုံးကို ဖော်ပြပါ။ ထို့အပါအဝင် ဇီဝဆိုင်ရာ စမ်းသပ်မှုများ (ဆဲလ်များကို အန်တီဖော်မ် ဖော်ပြခြင်း၊ အရေပြား အာရီးရှင်းဖော်ပြခြင်း၊ အရေပြားအတွင်း တုံ့ပြန်မှု စမ်းသပ်မှုများ) ကိုလည်း ဖော်ပြပါ။
သန့်စင်မှု နည်းလမ်းများကို ဖော်ပြပါ - သင်သည် အလေးချိန်မှုနည်းလမ်း (ရေငွေ့)၊ ETO ဓာတ်ငွေ၊ ဂမ္မာအလင်းရောင် သို့မဟုတ် E-beam ပိုးသတ်ခြင်းကို အသုံးပြုမည်လား။ ဤနည်းလမ်းများသည် ပစ္စည်းအများအပြားကို လိုအပ်ပါသည်။ ပြန်လည်အသုံးပြုနိုင်သော အယ်လ်ထရာဆောင်းဒ် ပရော့ဘ်ကြိုးများအတွက် ပစ္စည်းသည် အလေးချိန်မှုနည်းလမ်းဖြင့် ၁၀၀ ကြိမ် သို့မဟုတ် ထိုထက်ပိုများသော စက်ဝိုင်းများကို ခံနိုင်ရည်ရှိရပါမည်။
ပစ္စည်းဆိုင်ရာ စာရွက်စာတမ်းများ လိုအပ်ပါသည်။ သင်၏ ပေးသွင်းသူများသည် ပစ္စည်းအကုန်လုံး၏ ဖော်ပြချက်များ၊ ဇီဝသ совместимость လက်မှတ်များနှင့် ပိုးသတ်ခြင်း အတည်ပြုခြင်း အစီရင်ချက်များကို ပေးပေးရန် လိုအပ်ပါသည်။
ဤသည်များကို Hotten အတွက် စာရွက်စာတမ်းများမျှသာမက ကျွန်ုပ်တို့မှ ထုတ်လုပ်သော RF Ablation Cable နှင့် Dental Sensing Cable တိုင်းတွင် လူနေမှုအေးချမ်းရေး၏ အခြေခံအုတ်မြစ်ဖြစ်ပါသည်။
ပတ်ဝန်းကျင်နှင့် အသုံးပြုမှုအလိုက် အခြေအနေများကို အကဲဖြတ်ပါ။
အခြေခံလျှပ်စစ်နှင့် ယန္တရားဆိုင်ရာ အရည်အသွေးများအပြင် သင်၏ ကိရိယာကို အသုံးပြုမည့် အထူးအခြေအနေများကို စဉ်းစားပါ။
ဓာတုပစ္စည်းများနှင့် ထိတွေ့မှု - ကြိုးသည် သန့်စင်ရေးအေဂျင့်များ၊ ခန္တာကိုယ်အရည်များ သို့မဟုတ် အေးစက်ခွဲစိတ်ရေး အဆီများနှင့် ထိတွေ့မည်လား။ အထူးသဖြင့် ပိုမိုမှုန်းမှုများသော ပိုးသတ်ဆေးများကို အသုံးပြုရသည့် Surgical Scalpel Cables နှင့် Endoscope Cables တို့အတွက် ဓာတုပစ္စည်းများနှင့် ခံနိုင်ရည်ရှိမှု လိုအပ်ချက်များကို ဖော်ပြပါ။
အပူခါးသည်းခံနိုင်မှု အကွာအဝေး- အောက်ထရာကလေးဗ် စတီရီလိုက်ဇေးရှင်းအတွက် ပစ္စည်းများသည် ၁၃၅°စီ အထက်တွင် အလုပ်လုပ်နိုင်ရန် လိုအပ်ပြီး ကရိုင်ယိုအဘေလေးရှင်းအတွက် အလွန်နိမ့်သော အပူခါးများ လိုအပ်နိုင်ပါသည်။
ကွန်နက်တာ ပေါင်းစပ်မှု- ကြိုတင်ပြီးစုံလင်သော ကွန်နက်တာများ၊ အော်ဝာမော်လ်ဒ် စိတ်ဖိစီးမှု လျော့ချရေးများ သို့မဟုတ် အထူးပြုထားသော ဘက်ရှယ်များ အနက်မှ ရွေးချယ်ပါ။ ကွန်နက်တာ-ကြိုး အဆက်အသွယ်သည် အားနည်းသော နေရာဖြစ်လေ့ရှိပါသည်- ထိုနေရာကို ကောင်းစွာ ဒီဇိုင်းရေးဆွဲပါ။
နောက်ဆုံးဖြစ်သော်လည်း အလွန်အရေးကြီးသော အချက်မှာ ဆေးဘက်ဆိုင်ရာ အသုံးပြုမှုများအကြောင်း အသိပုံပုံရှိသော ထုတ်လုပ်သူနှင့် ပူးပေါင်းဆောင်ရွက်ခြင်းဖြစ်ပါသည်။ ဟော့တန်တွင် သင့်၏ အင်ဂျင်နီယာအဖွဲ့နှင့် ကျွန်ုပ်တို့၏ ပူးပေါင်းဆောင်ရွက်မှုသည် ကုသမှုဆိုင်ရာ လိုအပ်ချက်များကို ထုတ်လုပ်နိုင်ပြီး ယုံကြည်စိတ်ချရသော ကြိုးစီးရီးများအဖြစ် အကောင်အထည်ဖော်ရန် ဖြစ်ပါသည်။ ရှင်းလင်းပြီး အပြည့်အစုံသော အသေးစိတ်အချက်အလက်များဖြင့် သင့်၏ ဆေးဘက်ဆိုင်ရာ ကိရိယာကို ဈေးကွက်သို့ မိတ်ဆက်ရာတွင် မပေါ်လွင်သော စွမ်းဆောင်ရည်၊ ဘေးကင်းမှုနှင့် သက်တမ်းကို အာမခံပေးနိုင်မည့် အင်တာကောန်နက် ဖြေရှင်းနည်းကို သင်သည် သေချာစွာ ရရှိမည်ဖြစ်ပါသည်။
 အရေးကြီးသော သတင်းများ
အရေးကြီးသော သတင်းများ2025-12-17
2025-12-11
2025-12-05
2025-04-29